
by Prakash Achaary | Oct 19, 2022 | Blog
Electronic Clinical Outcome Assessments (eCOA) and Electronic Patient Reported Outcomes (ePRO) are innovating the clinical trial industry. It’s time for Sponsors to embrace accurate and efficient real-time electronic data collection directly from patients, in the...

by Prakash Achaary | Oct 3, 2022 | Blog
CANTON, Mich. (10/2/2022) – Data anonymization and document anonymization in clinical trial data is now more important than ever. In recent years, clinical trial data sharing has become a requirement as part of the regulatory process for EMA and Health Canada. ...
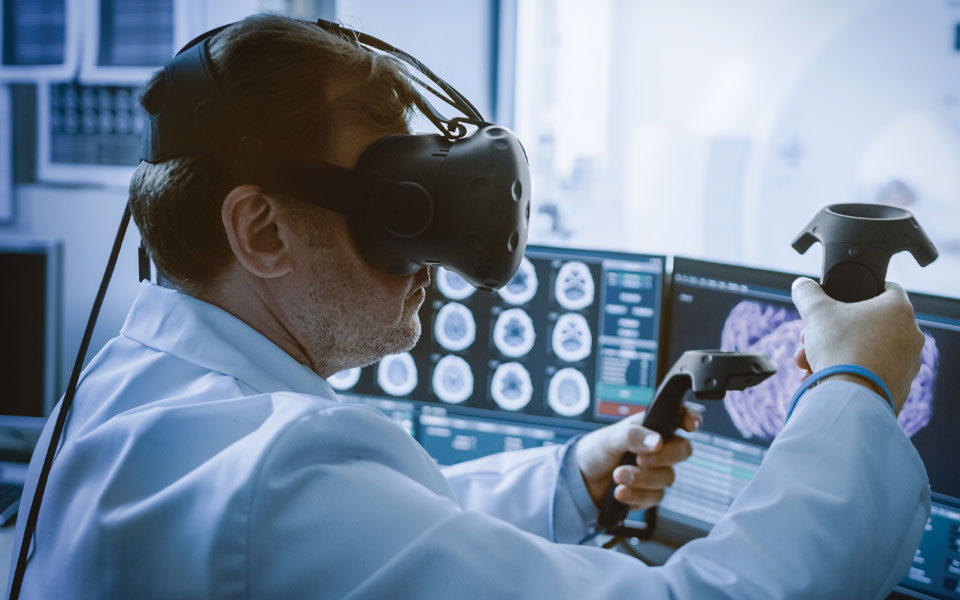
by Prakash Achaary | Sep 20, 2022 | Blog
Augmented reality (AR) and virtual reality (VR) medical devices are being integrated into the medical field at an accelerating rate. On July 12th and 13th, 2022, the FDA held a Patient Engagement Advisory Committee (PEAC) meeting to consider the capabilities, risks,...

by Prakash Achaary | Sep 15, 2022 | Blog
On 31 January 2022, the EU Clinical Trial Regulation 536/2014 (commonly known as EU-CTR) replaced the long-standing directive, the European Union (EU) Clinical Trial Directive 2001/20/EC (commonly known as EU‑CTD). There are important changes in the EU-CTR, compared...

by Prakash Achaary | Aug 29, 2022 | Blog
Consultant – a person who provides expert advice professionally. Drug discovery and development is a team endeavor with significant expertise needed at every stage. This expertise is a challenge for any company to cover completely in-house. With drug discovery and...






