rare disease therapeutic expertise
Experts at MMS are pioneers in rare disease and orphan drug applications
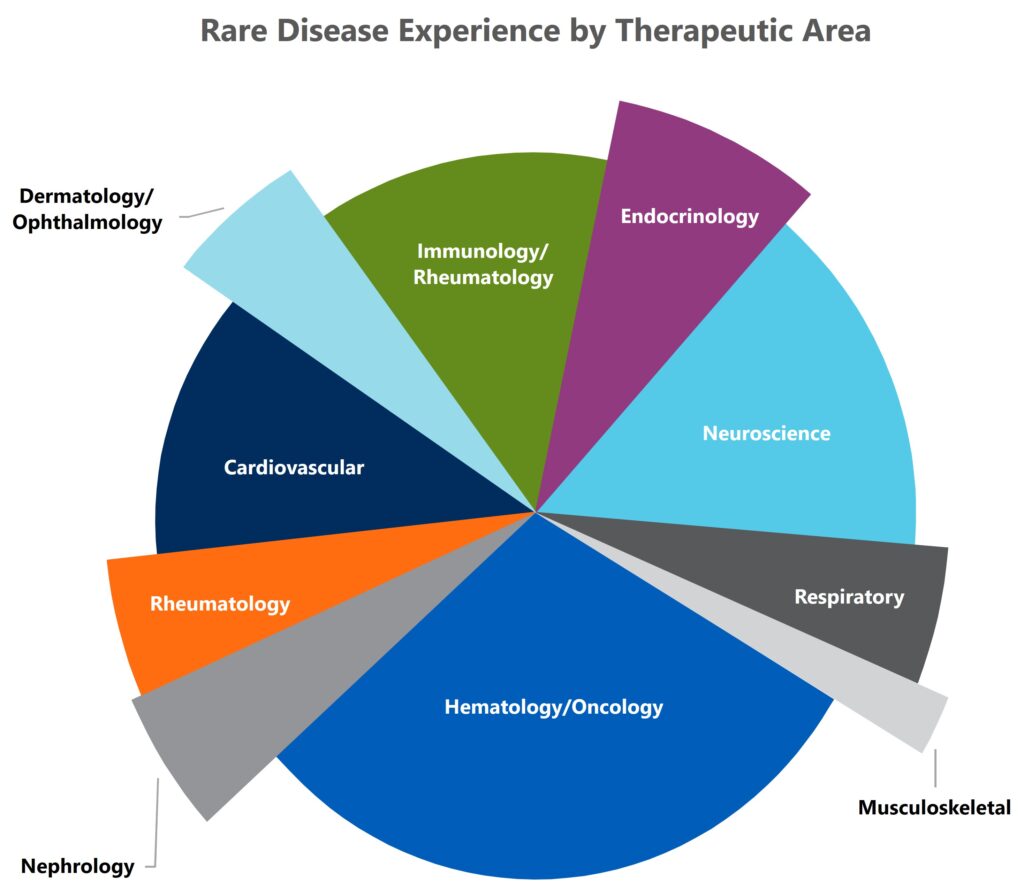
MMS has extensive experience and knowledge across various rare disease indications from pre-IND through post-marketing support.
MMS understands the importance of a customized regulatory strategy and the value of each data point when developing a therapeutic for rare disease indications. A data focused approach is needed given the impacts of having a limited number of patients for program development. Experts at MMS have led rare disease indications in a variety of therapeutic areas from the early planning IND/CTA stages through marketing applications for approval.
MMS rare disease therapeutic experience extends to the following areas:
Services Provided
- Regulatory strategy for orphan drug programs
- Agency meetings, including preparation of briefing materials and meeting attendance
- Orphan applications and annual report preparation, including literature search/summarization and the use of alternate data sources to show disease prevalence
- Clinical and statistical review of data supporting rare disease indications, including non-traditional sources like longitudinal or EMR data
Rare Disease Indications
We continue to work on many additional rare disease indications, adding to this list every day. To find out more about a specific TA or indication, please contact us.
- Duchenne Muscular Dystrophy
- Myasthenia Gravis
- Doose Syndrome
- Prader Willi Syndrome
- Sickle Cell Anemia
- Thymidine Kinase 2 Deficiency
- Still’s Disease
- Fetal and Neonatal Alloimmune Thrombocytopenia (FNAIT)
- Chronic Atypical Neutrophilic Dermatosis with Lipodystrophy and Elevated Temperature (CANDLE)
- Clear Cell Sarcoma
- Many others
Regulatory Knowledge
- Establish strong relationships and clear communication with the FDA, paving the way for flexibility given the unique challenges of rare disease programs
- Provide a rationale for the use of biomarkers in early phase investigations
- Recommend innovative trial designs and strategies to address low patient numbers
- Innovative strategies to address limited understanding of disease pathology and progression, variability in disease presentation, and a lack of established endpoints
- Advise and guide strategy for non-clinical research and data presentation
Fill out the form at right to speak with a rare disease expert
Find out more about our commitment to innovation and tech-enablement here
PRO-BONO ULTRA-RARE DISEASE REGULATORY ASSISTANCE PROGRAM CREATED BY MMS
Full Operational Support for Rare Disease Therapies
 Support for a rare disease pipeline leads the sponsor team to a successful early program milestones including an orphan drug designation and rare pediatric disease designation for their product.
Support for a rare disease pipeline leads the sponsor team to a successful early program milestones including an orphan drug designation and rare pediatric disease designation for their product.
Get in touch

